- Hits: 3204
Aspirin production

MEGBI-PLANING-COST-TIMLEINE-13/10/2023

Note: in step 11 and 17 we used normal filtration not vacuum filtration

Procedure of Aspirin Production (2nd Method)
Part Ⅰ: Preparation of the Aspirin
- Weigh 1 gram of salicylic acid and place it into a 50 ml Erlenmeyer flask.
- Add 3 ml of acetic anhydride to the flask. (under the hood)
- Add 3 drops of 85% Phosphoric acid and a magnetic stirring bar in the flask.
- Fill a beaker to the middle with water and place it on the heater to create a hot water bath.
- Set the flask in the water bath and secure it to the ring stand using clamps.
- While waiting for the water to boil, add 3 ml of water into 2 tubes and place them on an ice bath to let them chill.
- When the water bath starts boiling, start timing the reaction. When the mixture reacts at temperature 100℃ for 15 minutes, then the reaction is complete.
- Add 1 ml of distilled water to the flask after the reaction is complete.
- Turn off the heat and carefully remove the flask from the water bath. Add 9-10 ml of distilled water to the flask and mix it. As the flask cools, crystals of aspirin start to form.
- When the crystals are observed, place the flask in an ice bath for 10 minutes. If no crystals appear, scratch the bottom of the flask gently with the stirring rod.
- Set up the vacuum filtration and place an accurately sized filter paper on its funnel making sure it is covering all the holes.
- Turn on the vacuum filtration and wet the filter paper with distilled water, it should go through the funnel immediately.
- Add the aspirin from the flask on the filter paper. After the water has passed through the funnel, add one of the 3ml water tubes cooled earlier to rinse the aspirin.
- When all the water passes through the funnel, repeat the rinsing with second 3 ml tube. Then leave the vacuum filtration on for a couple minutes to help dry the aspirin.
Part Ⅱ: Recrystallization of the Aspirin
- Set a small amount of crude aspirin obtained aside for further testing later.
- Transfer the aspirin into a 50 ml Erlenmeyer flask, and add 4 ml of ethanol then warm it until the solid dissolves. Then immediately remove it from the heat and gradually add 13 ml of cold water to the flask. Crystals should form. Then place the flask on an ice-water bath. Then repeat the vacuum filtration process.
- Place the aspirin on the watch glass and leave it in a dry place for 24 hours.
- After it is dry, weigh the aspirin and note its weight.
Part Ⅲ: Purity of Aspirin
- Using the melting point apparatus, measure the melting point of crude aspirin and recrystallized aspirin. Record the melting point as a range.
- The FeCl3 test: get 4 tubes and place 1 drop of ethanol and 2 drops of FeCl3 in each tube. Add salicylic acid in the 1st tube, crude aspirin in the 2nd tube, recrystallized aspirin in the 3rd The 4th tube is control hence nothing is added.
- Shake each tube and observe the results by color change of each tube.
Results:
16/8/2022

Dry crystals of aspirin took it from filter paper after the filtration process

Quantity of crystal aspirin in gram

Results of qualitative test of FeCl3 with salicylic acid (purple) and aspirin (not purple)

Aspirin powder
Lab scale for aspirin production Poster.pptx
Another trial for aspirin production(22/11/2022)
In order to avoid any obstructive problems that may appear in pilot plant scale production , we repeated this trial(same procedure).
The experiment shows that there is a need to increase the time of crystallization with mixing for about 20 min so that the yield increases
(in comparison with the last trial where the time of crystallization was 15 min without mixing).
Here are the results after drying, weighing and FeCl3 testing :
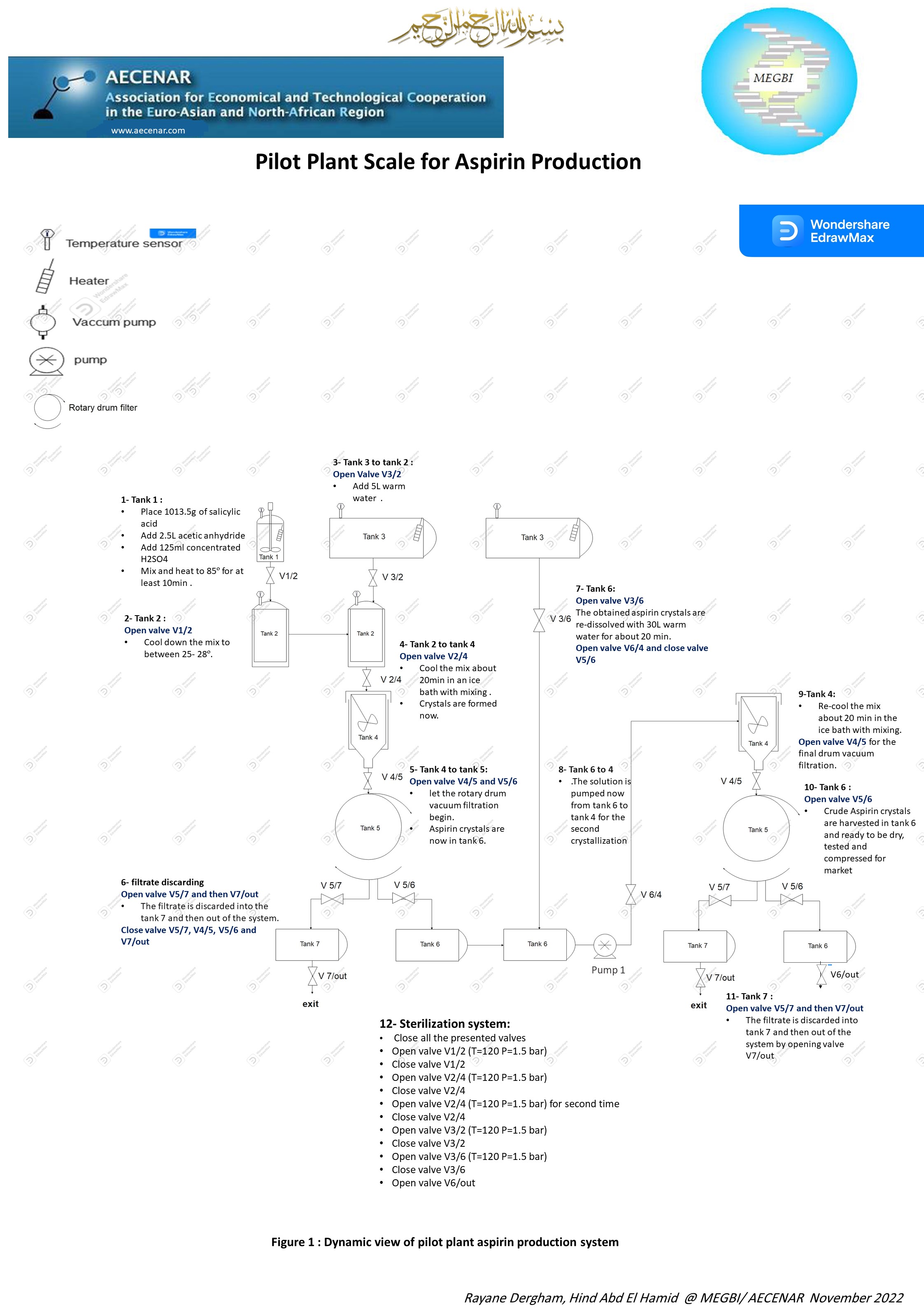
Pilot Plant Scale Aspirin Production

1-12-2022 Poster Pilot Plant scale for aspirin production
28-12-2022 Aspirin pilot plant
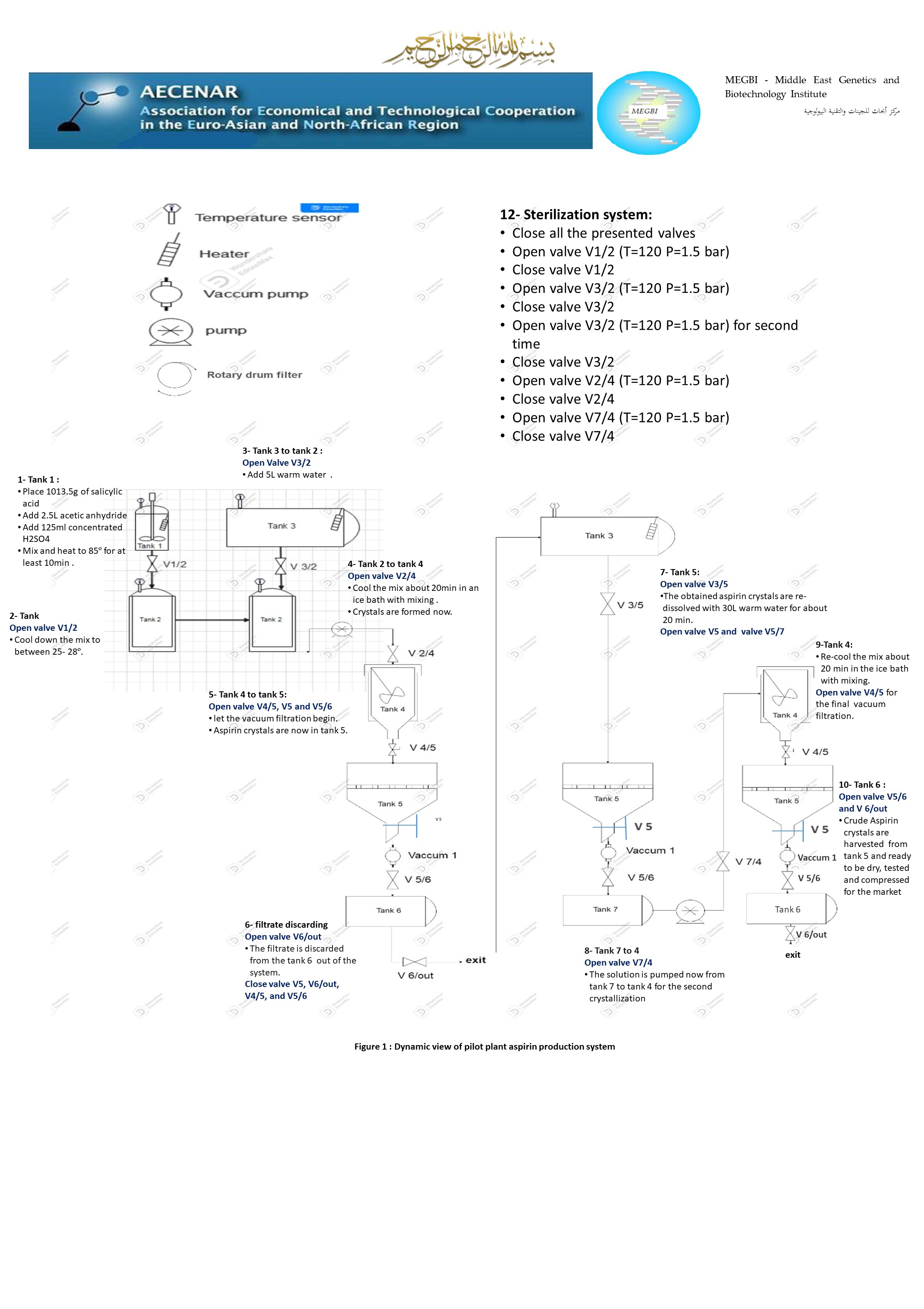
28-12-2022_Dynamic view of pilot plant scale aspirin production.pptx
06-12-2022_Dynamic view of pilot Plant Scale aspirin production with vacuum drum filter.pptx